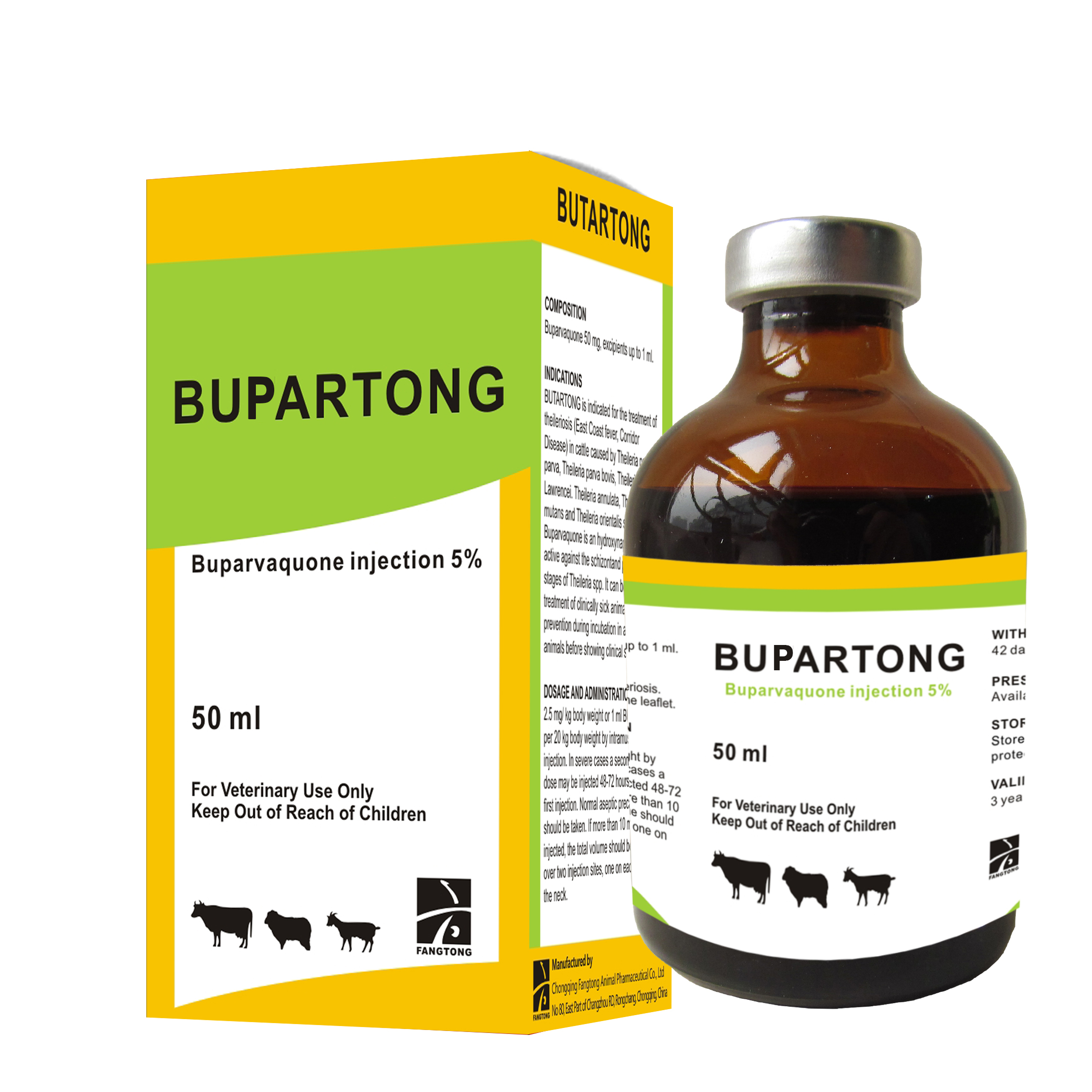
Presentation
Butalex is a clear, ruby-red solution for intramuscular injection. Each 1 ml contains 50mg buparvaquone.
Indications
Butalex is indicated for the treatment of theileriosis (East Coast fever, Corridor Disease, Tropical theileriosis etc.) in cattle caused by strains of Theileria parva (parva), T. parva (bovis), T. parva (lawrence), T, Mutans, T. annulata and T. orientalis (sergenti).
It is active against both the schizont and piroplasm stages of Theileria species and may be used in the incubation period of the disease or when clinical signs are apparent. Treatment should be given to clinically affected animals and to in-contact animals which may be infected but not showing clinical signs.
Dosage and Administration
Dose
A single injection of 2.5mg per kg (1 ml Butalex per 20kg bodyweight) is usually sufficient.
In severe cases, a further treatment with Butalex, at the same dose of 1 ml Butalex per 20kg bodyweight (2.5mg buparvaquone per kg) may be required. Normally this should be given within 48-72 hours of the initial injection.
Administration
Intramuscular injection into the muscles of the neck is recommended observing normal aseptic precautions. Care should be taken to avoid injection through wet or dirty areas of skin. No more than 10ml should be injected into a single site. When the dose volume exceeds 10ml the required dose should be split and injected into separate sites in the neck. In severe cases where a second treatment is necessary, this should be injected at a separate site, preferably on the opposite side of the neck.
Warnings and Precautions
Butalex must not be given by intravenous or subcutaneous injection.
Side effects
Localised, painless, oedematous swelling may occasionally be seen at the injection site.
Protection and Consumers
Milk taken from lactating animals during the 48 hours immediately following treatment with Butalex injection should not be used for human consumption.
Animals should not be slaughtered for human consumption within 42 days of the last treatment.
Further Information
In case of severe theileriosis relapses may occur, requiring further treatment within Butalex.
Recovered animals will develop homologous immunity. However any subsequent infections within antigenically unrelated species or strains of Theileria may require further treatment with Butalex.
In the presence of an outbreak of theileriosis, prompt dipping or spraying of all animals at risk must be an intergral part of the disease control procedure. Care should be taken to ensure that an approved acaricide is used, at the correct concentration and that all animals are dipped or sprayed. Sick animals should be hand sprayed, causing as little stress as possible. Dead animals should be hand sprayed, as any ticks which leave them may pass on the infection.
Attention should also be given to the possibility of other tick-borne infections. The clinical signs of these diseases may have been masked by those of theileriosis or latent infections may become patent as the animal’s immune response is compromised.
Babesiosis and anaplasmosis will require specific therapy with Imizol?
Supportive antibacterial chemotherapy may be needed for the control of secondary infection.
Attention to nutrition is recommended as an aid to convalescence. Severely affected animals should not be subjected to stress, e.g. walking over long distances, dipping etc.
Anaemia is a frequent characteristic of Theileria spp infections, particularly those caused by T. annulata, T. sergenti and T. mutans.
Consideration should therefore be given to the need to give supportive therapy for this anaemia.
Theileriosis has severe depressant effects on the immune system. Therefore, it is recommended that any vaccinations be delayed until the animal has recovered.
Storage Recommendations
Store below 25℃.
Protect from light.