ABSTRACT
Twelve healthy desert sheep were injected intramuscularly (IM) with multiple doses of 5 g kg-1 oxytetracycline (OTC) for detection of OTC residues in different tissues (Liver, kidney, muscle and site of injection). The animals were slaughtered and the tissue samples were collected from each sheep at times 1, 3, 7 and 10 days after drug administration. Samples were processed using microbiological method. The inhibition zone diameters were interpreted with oxytetracycline standard curve to obtain tissue concentrations. Oxytetracycline concentrations were evaluated in tissue at different times. High concentration were noted in liver (105±10.91 μg g-1) at day 1, kidney (234.93±10.57 μg g-1) at day 3 and muscle (3084.20±227.10 μg g-1) at day 1, while the highest concentrations were observed at site of injection (5963±227.10 μg g-1) at day 1. Day 7 and 10 showed low concentration of OTC in the liver (2.51±0.1 μg g-1), kidney (6.70±0. 1 μg g-1) and muscle (70.86±0.6 μg g-1). This study indicates the presence of long acting OTC concentrations in various tissues after injection of the drug. Significant difference in concentrations of the drug in the various tissues studied was demonstrated.
INTRODUCTION
Oxytetracycline is a highly active, broad-spectrum antibiotic which is widely used in veterinary medicine for the treatment of respiratory and gastrointestinal infectious disease. It is active against aerobic Gram-positive and Gram-negative bacteria, Rickettsia, Mycoplasma and Chlamydia (Rivier and Spoo, 1995). Oxytetracycline is produced by a fermentation process by Actinomycete, Streptomyces Rimosus. FAO (1991) reported that OTC was available in four major dosage products for animal health such as feed premix, soluble powder, injectable solutions and tablets. Oxytetracycline has been well tolerated when administered orally or by the intramuscular (IM) route as an aqueous solution. Oxytetracycline passed the blood brain barrier, placental, intestinal, serous membrane and milk barriers and was excreted mainly by urinary and biliary systems. It has a low toxicity and the Maximal Residual Levels (MRLs) was 100 μg g-1 for muscle, 300 μg g-1 for liver, 600 μg g-1 for kidney and 100 μg g-1 for milk (WHO, 1996).
Many public health problems may arise from OTC residues. These include drug allergy, hypersensitivity, skin rashes and phototoxic dermatitis (Dewdney, 1977). Depaola (1995) reported that, the presence of OTC residues in feed may increase the bacterial resistance. The binding of tetracycline to calcium may result in inhibition of teeth development and skeletal growth (WHO, 1996). The detection of drug residues in food of animal origin were performed by using specific chemical and microbiological methods (Cherlet et al., 2003) according to physico-chemical or biological properties of the substance to be detected. The aim of the experiments presented here was to determine the concentration levels of OTC in certain organs after IM administration in desert sheep.
MATERIALS AND METHODS
Animal: Twelve male desert sheep, weighing between 25-35 kg and aged 9-12 month were used. They were housed in pens (3×3 m) and fed with balance diet (concentrate and forage) and water was available ad libitum. After two weeks adaptation period, maximum therapeutic dose of oxytetracycline (Alamycin LA, Norbrook laboratories limited station work, Newry, North Ireland) was given intramuscularly (IM) at dose of 5 g kg-1 daily for 5 days, the animals were slaughtered and tissue samples (liver, kidney, muscle and site of injection) were collected in sterile plastic bags at 1, 3, 7 and 10 days after the last administration and frozen at -20°C for drug assay by microbiological method using Bacillus subtitilis BGA spores (DSM618) (Gorman et al., 1983).
According to (Koenen-Dierick et al., 1995) the logarithm concentration of OTC of known concentrations were plotted versus mean inhibition zone diameters. From calibration curve the concentration of OTC in samples were calculated.
Detection of oxytetracycline residues in tissue samples: To detect antibiotic quantity in tissue samples. One gram from liver, kidney, muscle and site of injection was prepared using sterile corkborer, blades and a balance in aseptic conditions and placed on the plates and incubated over night at 30°C for measuring the diameter of inhibition zone of each tissue. The inhibition zones measurement was compared with a calibration curve for the quantity of residues as described by Koenen-Dierick et al. (1995) and Koenen-Dreick and de Beery (1998).
Statistical analysis: Statistical analysis of the data obtained was carried out using ANOVA with significance difference p<0.05.
RESULTS
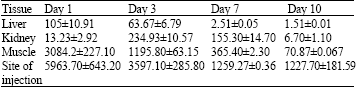
The positive results showed the inhibition activities around the tested tissue while negative results showed no growth. The comparison of inhibition zone diameter of the positive samples with the calibration curve of standard OTC concentrations showed tissue samples concentrations. The concentrations were detected in liver, kidney, muscle and site of injection (Table 1).
Liver concentration significantly decreased from 105±10.91 to 63.67±6.79, 2.51±0.05, 1.51±0.01 μg g-1 at day 1, 3 and 7, respectively.
Kidney showed the lowest concentrations (13.23±2.92 μg g-1) at day 1 while the highest concentrations (234.93±10.57 μg g-1) were observed at day 3. Then the concentrations decreased until it was disappeared (Table 1).
Muscle concentrations ranging from 3084.20±227.10 to 70.87±0.06 μg g-1. The high concentration was found at day 1 while day 10 showed concentration at the level of 70.87±0.06 μg g-1 (Table 1).
| Table 1: | Tissue residues (μg g-1) of oxytetracycline after intramuscular administration of 5 g kg-1 for 5 days to twelve healthy sheep |
 |
|
| All values are Mean±SEM | |
Mean OTC residue levels for site of injection decreased from 5963.70±643.20 to 1227.70±181.59 μg g-1 at day 1 and day 10, respectively. The greatest concentrations of OTC were observed at the site of injection at day 1 then the concentration gradually decreased but significant amount of the drug persisted up to 10 days (Table 1).
DISCUSSION
The present data clearly demonstrates that oxytetracycline residues appeared at the first day after the last injection in liver, kidney, muscle and site of injection.
The concentration of OTC following a therapeutic dose were higher than the Minimum Residue Limits (MRLs) suggested by FAO (1995) and WHO (1995a and b). These results were similar to those reported from individual organs in slaughter houses (Muriuki et al., 2001) using HPLC method.
Similar observations have been described for OTC in hens (Serrano et al., 1999) where the highest concentration was found in kidney followed by liver using HPLC method (Samanidou et al., 2005). The finding that liver and kidney were containing residues of antibiotic may be due to the fact that these organs are concerned with elimination of the drug.
The highest levels of OTC were found in site of injection. This is in agreement with previous reports for OTC residues (Craigmill et al., 2000). The concentration was highest in sheep for the injection site, followed in decreasing order by the kidney, liver, muscle and fat using HPLC method. However, WHO (1991) reported that the high level of residues only persisted at injection sites. These results suggest that the absorption of the drug was slow from site of injection. Furthermore, the occurrence of highest concentration of drug in the circulation at 3 days post-injection would substantiate this observation. These observations agree with the studies of OTC long-acting formulations reported by Hall et al. (1989) and De Banting and Baggot (1996).
Present finding were different from that of Limpka et al. (1993) who found that 82% of OTC is excreted from tissue at 48 h post dosing after IM injection, the difference could be due to the species of the animal, dosage or formulation.
Send your message to us:
Post time: Mar-11-2020

